The Spike Protein and Blood and Endothelial Cells
Sialylated Glycan Bindings from SARS-CoV-2 Spike Protein to Blood and Endothelial Cells Govern the Severe Morbidities of COVID-19
TLDR: An AI Summary of a recent paper exploring spike protein damage through its interactions with blood and endothelial cells. Interesting to see how the AI is now finding the spike-vaccine connections. Also interesting was the luteolin connection, which I have previously talked about here and here. I take Oregano (dried leaves ground in a coffee grinder mixed with my food) as a high luteolin-content food (it works out at about 5mg of luteolin per $1 of dried herb. However, the cheapest way to buy luteolin is as a supplement, where 100mg can be bought for as little as 20 cents).
AI summary:
The paper titled "Sialylated Glycan Bindings from SARS-CoV-2 Spike Protein to Blood and Endothelial Cells Govern the Severe Morbidities of COVID-19" delves into the biochemical interactions between the SARS-CoV-2 spike protein (SP) and host cells, and how these interactions contribute to the severe morbidities observed in COVID-19. Here is a summary of the key findings:
SARS-CoV-2 SP and Red Blood Cell (RBC) Aggregation: The SARS-CoV-2 SP has been shown to attach and aggregate RBCs, causing pulmonary and extrapulmonary microthrombi and hypoxia in severe COVID-19 patients.
Clinical Observations of Microvascular Occlusion: COVID-19 patients often exhibit microvascular occlusion in various organ systems, accompanied by morbidities like intravascular clotting and peripheral ischemia.
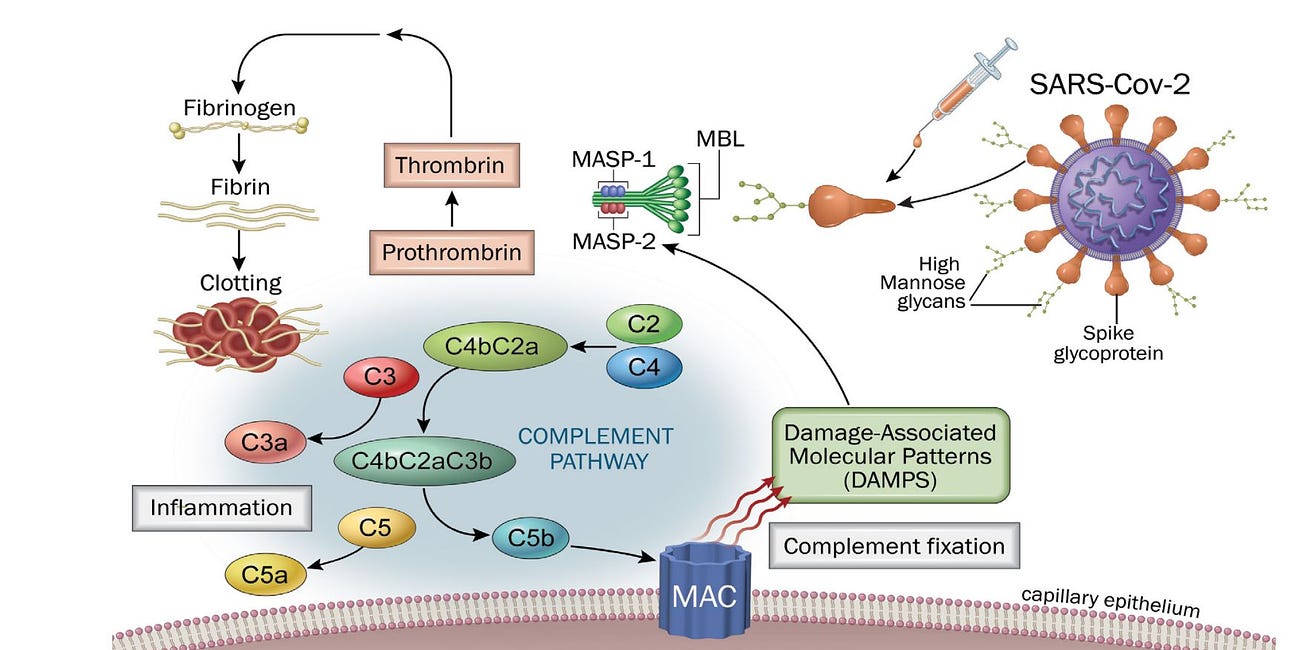
Glycans with Sialic Acid and Host Cell Interaction: The SARS-CoV-2 virus relies less on the ACE2 receptor for cell entry and more on glycans with sialic acid (SA) terminal moieties found on the SP and host cells, such as RBCs, platelets, leukocytes, and endothelial cells.
In Vitro and In Vivo Evidence of RBC Aggregation by SARS-CoV-2 SP: Multiple studies have demonstrated that SARS-CoV-2 SP causes RBC clumping, or hemagglutination, in various settings, including human RBCs and zebrafish embryos, suggesting a mechanism for observed blood flow reductions and thrombosis.
Presence of SARS-CoV-2 SP in Patients' Blood: SARS-CoV-2 SP and its subunits have been found in the blood of COVID-19 patients, with persistent presence in some cases post-infection, indicating the potential of SP leakage outside infected cells during viral replication.
Evidence of RBC Aggregation in COVID-19 Patients: Clinical studies have documented blood cell clumping and the presence of larger RBC aggregates in COVID-19 patients, correlating with enhanced blood coagulation.
Glycan Bindings and Endothelial Damage: Attachments of SARS-CoV-2 SP to platelets and endothelial cells cause endothelial damage, platelet activation, and coagulation, contributing to severe COVID-19 morbidities.
Experimentally Induced RBC Clumping and Parallels to COVID-19: Experimental studies have shown that high-molecular-weight dextran can induce RBC clumping similar to what is observed in severe COVID-19 cases, causing microvascular occlusion and tissue damage.
Corresponding Morbidities in Severe COVID-19: The morbidities caused by experimentally induced RBC aggregation, such as microvascular occlusion, hypoxia, arterial and venous thromboembolisms, and multiorgan damage, have been commonly observed in severe COVID-19 cases.
Redistribution of Blood Flow in COVID-19 Patients: COVID-19 patients, especially those with long COVID, show a marked reduction in the density of functional capillaries, indicating impaired microvascular health.
Major Risk Factors Correlating with RBC Aggregation: Age, diabetes, and obesity, major risk factors for severe COVID-19, correlate with increased RBC aggregability. This increased aggregability, coupled with slower blood flow in older individuals, contributes to a higher rate of microvascular occlusion.
SARS-CoV-2 SP Inducing Microvascular Occlusion: Studies have shown that SARS-CoV-2 SP, even unattached to the whole virus, can cause microvascular occlusion, myocardial damage, and other cardiovascular complications.
Clinical Signs of Microvascular Occlusion Post-Exposure to SARS-CoV-2 SP: Optical coherence tomography angiography has provided insights into microvascular occlusion in COVID-19 patients, especially after exposure to SARS-CoV-2 SP.
These findings highlight the crucial role of the SARS-CoV-2 spike protein in causing severe morbidities in COVID-19, particularly through its interaction with blood and endothelial cells, leading to widespread vascular and tissue damage.
Spike protein produced following mRNA vaccination could also contribute to cardiac issues using the same mechanisms:
Presence of Spike Protein Post-Vaccination: Some studies have found the presence of spike protein in the blood of individuals after mRNA vaccination, which is notable because it is the same protein produced by the SARS-CoV-2 virus itself.
Association with Myocardial Injury: Research has indicated that there might be a correlation between the presence of the vaccine-induced spike protein and myocardial injury. For example, one study found elevated levels of spike protein in the plasma of individuals with post-vaccine myocarditis, suggesting a potential link between the presence of the spike protein and cardiac issues.
Detection in Human Tissue Post-Vaccination: Another study assessing human tissues from autopsies of patients who died after vaccination found traces of the vaccine in certain tissues, including the heart, in some patients who had been vaccinated within 30 days of their death. These findings were associated with myocardial injury, although the cause of death was not directly linked to the vaccine.
Given what we know about the damaging effects of the spike protein, what do you suggest?
While specific treatments should always be discussed with a healthcare provider, some general strategies could be beneficial:
Nutrition and Hydration: A balanced diet rich in fruits, vegetables, whole grains, lean proteins, and healthy fats can support the immune system. Adequate hydration is also important for overall health.
Physical Activity: Regular physical activity can help maintain a healthy cardiovascular system, improve mood, and boost immune function. It's essential to tailor exercise intensity to individual health status and abilities.
Rest and Sleep: Adequate rest and good sleep quality are crucial for recovery and overall health. They play a key role in immune function and the body's healing processes.
Stress Management: Chronic stress can negatively impact immune function and overall health. Techniques such as mindfulness, meditation, yoga, or other relaxation practices can be beneficial.
Avoiding Harmful Substances: Avoiding smoking, excessive alcohol consumption, and other harmful substances is important for maintaining good health and supporting recovery.
Support Network: A strong support network of family, friends, or support groups can provide emotional support, which is important for mental and physical health.
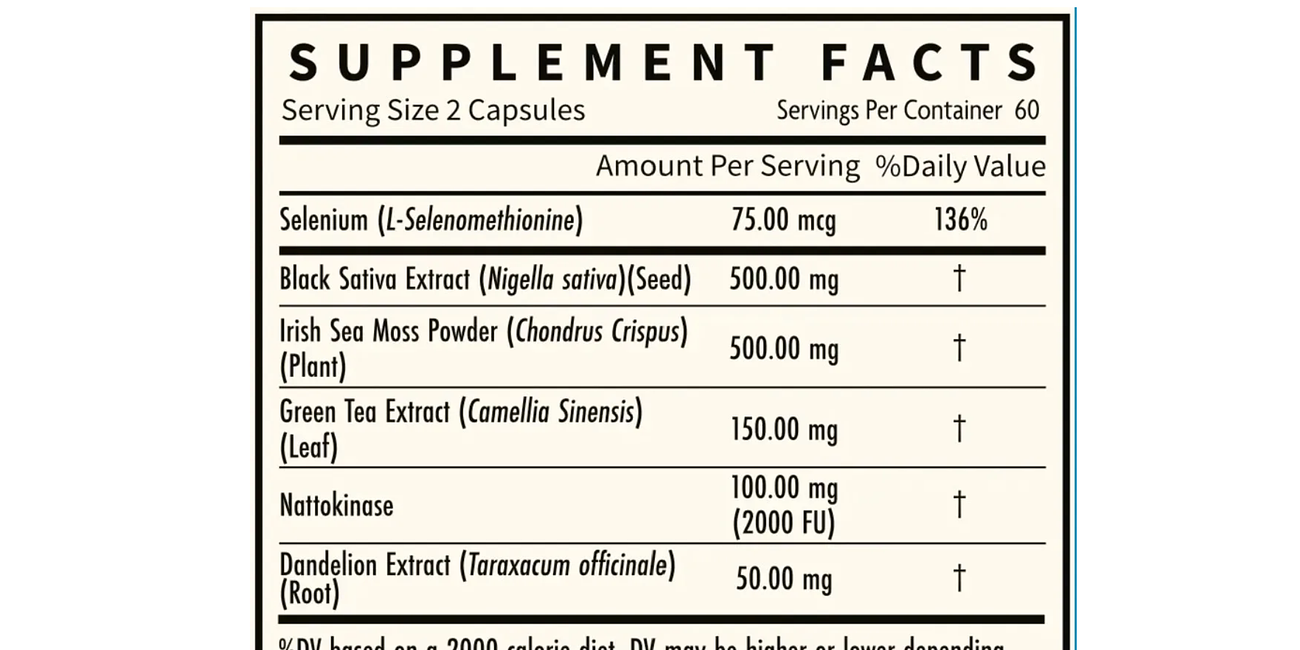
Supplements and Vitamins: Some may consider supplements and vitamins, but these should be taken under the guidance of a healthcare professional. There's no one-size-fits-all recommendation, as needs can vary greatly between individuals.
Remember, individual health situations vary, and what works for one person may not be appropriate for another
What supplements could be helpful?
To mitigate potential damage from the spike protein, whether due to COVID-19 infection or vaccination, some studies suggest the use of natural antioxidants. Polyphenols, for instance, are known for their antioxidant properties and may help counteract oxidative stress induced by the spike protein.
One specific compound mentioned is luteolin, which could be beneficial in this context.
The result is inflammation around brain blood vessels (22) that may generate a vicious cycle or autoimmune loop of neuroimmune processes casing L-COVID. In fact, preliminary reports suggest that mast cells may be involved in COVID-19 (47-49) and that individuals taking substances interfering with the mast cells or their mediators are less likely to suffer severe COVID-19 (33, 43). COVID-19 can also affect the hypothalamic-pituitary-adrenal (HPA) axis (50), which is typically activated by stress (51), but also has pro-inflammatory effects via stimulation of mast cells by corticotropin-releasing hormone (CRH) (53). Mast cells can interact with microglia in the brain, leading to their activation with subsequent neuroinflammation (52-54) and cognitive dysfunction (55). Microglia express Tolllike receptors (TLRs) (56), activated by damage associated molecular patterns (DAMPs) and were recently implicated in COVID-19 (57-60). Given the above, it is reasonable to consider preventing the detrimental actions of the spike protein (Table I). This could be accomplished by the use of liposomal formulations (61) of the natural flavonoids luteolin and methoxyluteolin (62-64), which are safe (65, 66) and have broad anti-viral properties (67) by acting at different steps (Fig. 1): Spike protein binding to its ACE2 receptor; 2) Activation of serine proteinases responsible for “priming” the Spike protein for entry into the cells; 3) Viral replication within the nucleus; 4) Activation of TLR7/8 found in the endosomes by single-stranded RNA viruses like SARS-CoV-2; 5) Production of proinflammatory cytokines. Furthermore, luteolin and methoxyluteolin better penetrate into the brain, inhibiting both microglia (68-70) and mast cells (71, 72), while they also reduce neuroinflammation (66, 73, 74) and cognitive dysfunction (75, 76), especially brain fog (77, 78).
While luteolin is one supplement mentioned for its potential benefits against oxidative stress related to the spike protein, there are other supplements that might also be helpful:
Vitamin C: Known for its antioxidant properties and immune support.
Vitamin D: Important for immune function and overall health.
Zinc: Plays a role in immune function and inflammation reduction.
Omega-3 Fatty Acids: Can help with inflammation and cardiovascular health.
N-acetylcysteine (NAC): An antioxidant that can support lung health and reduce oxidative stress.
I have spoken about luteolin before, and here is a quote from a paper:
A naturally occurring flavonoid, luteolin is 2-(3,4-dihydroxyphenyl)-5,7-dihydroxychromen-4-one. It is found in many vegetables (celery, parsley, broccoli, carrots, peppers, and cabbages); fruits (apple skins); flowers (chrysanthemum) and in medicinal herbs. It is a potent antioxidant, inhibits tumor cell proliferation and suppresses metastasis. It is also an anti-inflammatory agent and an immune system modulator (Lin et al., 2008; National Center for Biotechnology Information, 2020g). Luteolin exhibits a concentration-dependent inhibition of the production of TNFα and IL-1β in LPS/IFN-induced primary microglia and BV-2 microglial cells. This inhibitory effect results from an inhibition on NF-κB, STAT1 and IRF1; all essentially required for the transcription of pro-inflammatory genes (Kao et al., 2011). In another study, a significant inhibition of TNFα, IL-6, CXCL8, GM-CS and suppression of NF-κB activation was observed with luteolin treatment on PMA plus A23187-induced HMC-1 cells (Kang et al., 2010). In human monocytes under hyperglycemic condition, luteolin brought about a significant reduction in the release of IL-6 and TNFα by suppressing NF-κB activity (Kim et al., 2014). In a study with human whole blood incubated with LPS, luteolin effectively inhibited the production of IL-1β, IL-6, TNFα and IFNγ (Ribeiro et al., 2010). Primary murine microglia and BV-2 microglial cells stimulated with LPS exhibited significant increase in levels of inflammatory cytokine, IL-6. Pretreatment with luteolin was found to attenuate this increase in IL-6; both at mRNA and protein level. While luteolin was found to bring about a marked reduction in the binding activity of AP1 transcription factor and inhibit JNK phosphorylation, it had no significant effect on the LPS-induced increase in NF-κB DNA binding activity nor the LPS-induced IκBα degradation. Furthermore, the in vivo studies on mice treated with luteolin revealed that it successfully reduced the plasma levels of IL-6 and the mRNA levels of IL-6 in the hippocampus, but not in the cortex or cerebellum. This inhibitory activity of luteolin on the LPS-induced production of IL-6 can be attributed to its ability to inhibit the both JNK signaling pathway and the activation of AP1 in the microglia (Jang et al., 2008). Luteolin was also found to significantly reduce the serum levels of pro-inflammatory cytokines IL-1β, IL-6 and TNFα in MSU induced-inflammation in rats (Lodhi et al., 2019). Luteolin effectively inhibited the production of TNFα and IL-6 in LPS/INFγ stimulated RAW264.7 cells. NF-κB expression was also suppressed in a dose-dependent manner, following luteolin treatment (Lee et al., 2016). Luteolin pretreatment was found to bring a marked decrease in the mRNA expression and the release of IL-6, IL-8 and VEGF in a dose dependent manner in human HaCaT and primary keratinocytes on TNF stimulation. The TNF-induced phosphorylation, nuclear translocation and DNA binding of NF-κB was significantly brought down by treatment with luteolin. Furthermore, the TNF-induced mRNA expression of the NFKB1 and RELA genes which encode the two NF-κB subunits (NF-κB p50 and NF-κB p65, respectively) was also reduced by luteolin treatment, suggesting that luteolin reduces pro-inflammatory cytokine production by decreasing NF-κB induction (Weng et al., 2014). Luteolin also has broad anti-viral activity, inhibits mast cells and potentially inhibits SARS-CoV-2 main protease (3CLpro) as revealed in recent in docking studies (Theoharides, 2020). Taken together with its immunomodulatory properties, luteolin is an important natural agent for consideration as a potential therapeutic that can be further evaluated as an adjuvant to ameliorate the cytokine storm observed in COVID-19 and to potentially inhibit SARS-CoV-2 infection. Further clinical trails in this regard to ascertain the safety, efficacy and dosage for COVID-19 treatment needs to be conducted.
Further reading:















Forgive me but one ingredient, to help the entirety of Humanity, is missing: about a million bullets, for the politicians, scientists, doctors & pundits who funded, designed and released this Doomsday weapon.
About omega 3. Fish oil capsules often are rancid. Even if you keep it in the fridge- God only knows how long it was sitting on a shelf beforehand. I know avocados are a good source of omega 3, there must be a few other useful foods out there...